Year 9
Atomic structure and the periodic table
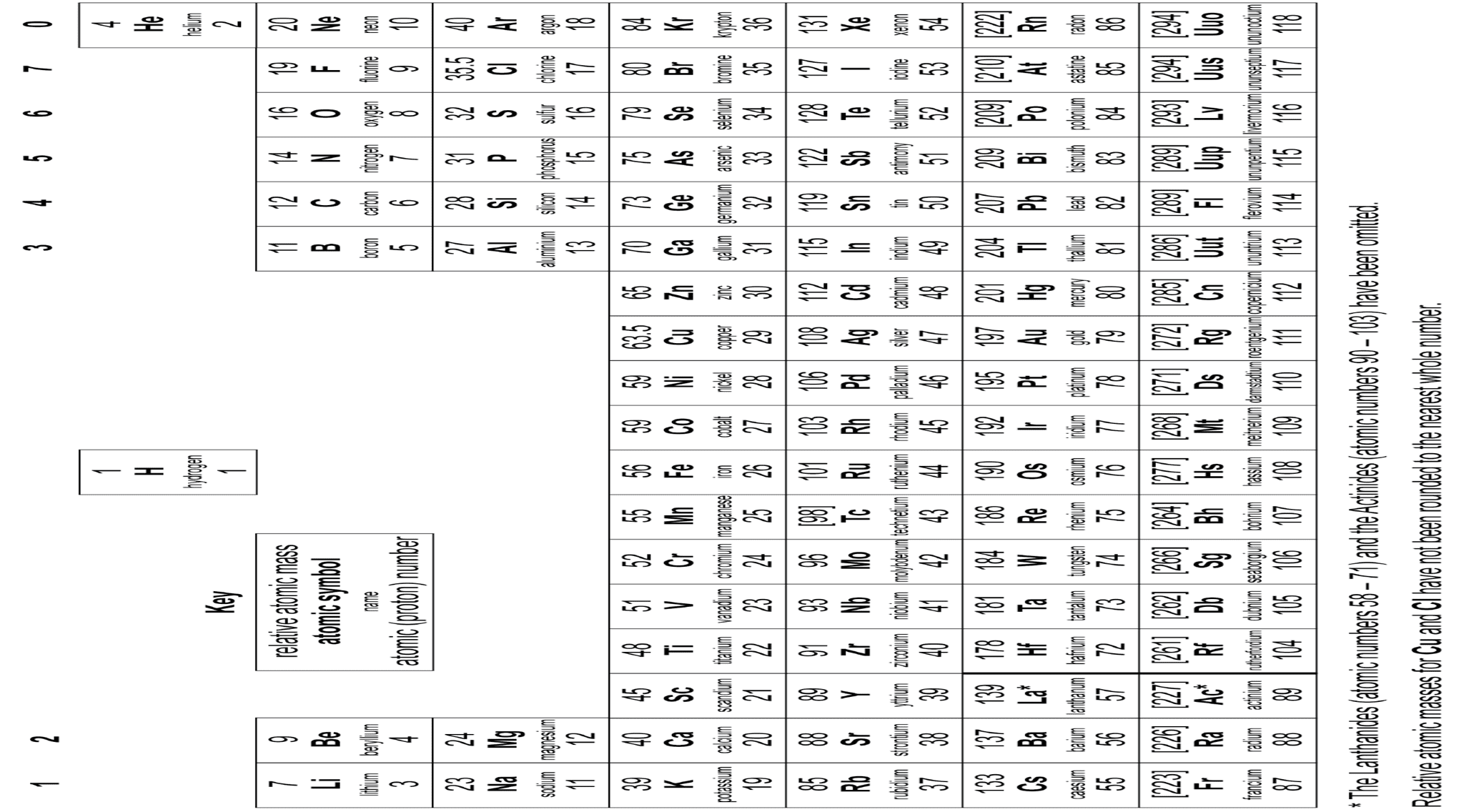
The periodic table provides you with a structured organisation of the known chemical elements from which you can make sense of their properties. You will study the arrangement of elements in the modern periodic table and how they can be explained in terms of atomic structure.
Read MoreBonding, structure, and the properties of matter
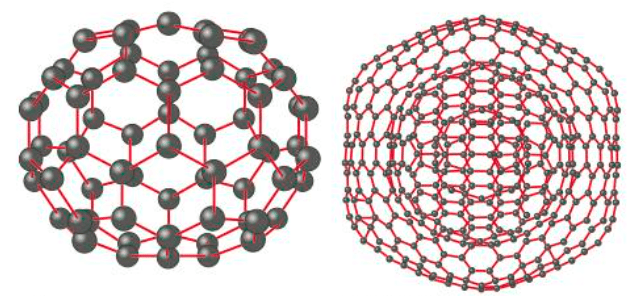
Chemists use theories of structure and bonding to explain the physical and chemical properties of materials. Analysing structures shows that atoms can be arranged in a variety of ways. Theories of bonding explain how atoms are
held together in these structures.
Year 10
Quantitative chemistry

You will use quantitative analysis to determine the formulae of compounds and the equations for reactions.
Identifying different types of chemical reaction allows chemists to make sense of how different chemicals react together, to establish patterns and to make predictions about the behaviour of other chemicals.
Chemical changes

You will study different types of chemical reaction, understanding how chemical changes can be predicted and used to make a variety of substances. You will learn in detail about acids and their reactions with various other chemicals.
Read MoreEnergy changes
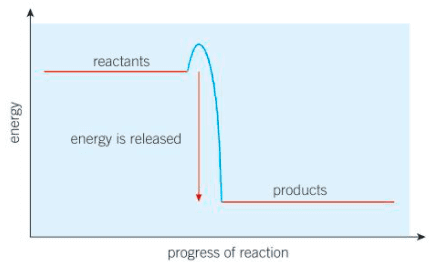
Energy changes are an important part of chemical reactions. The interaction of particles often
involves transfers of energy due to the breaking and formation of bonds. Some interactions between ions can result in the production of electricity.
The rate and extent of chemical change
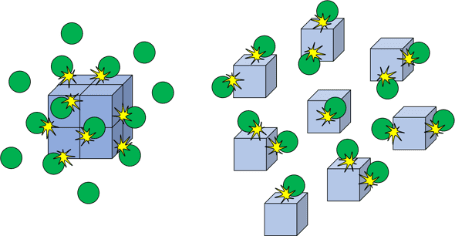
Chemical reactions can occur at vastly different rates. Whilst the reactivity of chemicals is a
significant factor in how fast chemical reactions proceed, there are many variables that can be
manipulated in order to speed them up or slow them down. Chemical reactions may also be
reversible and therefore the effect of different variables needs to be established in order to identify
how to maximise the yield of desired product.
Year 11
Organic Chemistry
The chemistry of carbon compounds is so important that it forms a separate branch of chemistry. A
great variety of carbon compounds is possible because carbon atoms can form chains and rings
linked by C-C bonds. This branch of chemistry gets its name from the fact that the main sources of
organic compounds are living, or once-living materials from plants and animals. These sources
include fossil fuels which are a major source of feedstock for the petrochemical industry.
Chemical Analysis

Analysts have developed a range of qualitative tests to detect specific chemicals. The tests are
based on reactions that produce a gas with distinctive properties, or a colour change or an
insoluble solid that appears as a precipitate.
Instrumental methods provide fast, sensitive and accurate means of analysing chemicals, and are
particularly useful when the amount of chemical being analysed is small.
Chemistry of the atmosphere

The Earth’s atmosphere is dynamic and forever changing. The causes of these changes are
sometimes man-made and sometimes part of many natural cycles. Scientists use very complex
software to predict weather and climate change as there are many variables that can influence this.
The problems caused by increased levels of air pollutants require scientists and engineers to
develop solutions that help to reduce the impact of human activity.
Using resources

Industries use the Earth’s natural resources to manufacture useful products. Pollution, disposal of waste products and changing land use has a significant
effect on the environment, and environmental chemists study how human activity has affected the
Earth’s natural cycles, and how damaging effects can be minimised.

